On 17-Nov-2022, The USA FDA approved Teplizumab. This is the first drug ever approved in the United States which changes the course of Type-1 Diabetes. This is a long blog which will discuss the approval from several different points of view.
As you read this blog remember that it is the first blog I've ever written on a treatment that has been approved for use in the US. I'm in new territory here; we all are. If you want an excellent (and shorter) overview of Teplizumab, I recommend this diaTribe article:
https://diatribe.org/fda-approves-tzield-teplizumab-delay-type-1-diabetes
Here is the official FDA announcement:
https://www.fda.gov/news-events/press-announcements/fda-approves-first-drug-can-delay-onset-type-1-diabetes
What Was Approved? What Will It Do?
First: This is not a cure for type 1 diabetes.
It was approved because it delayed the onset of T1D for an average of 2 years. Continued testing suggests the actual delay might be 3 years or even longer. However, the current information is that this drug delays the onset of T1D. It is not being approved to prevent or to cure T1D.
This drug was approved only for people who are 8 years old or older and have stage 2 of type 1 diabetes. Other uses are "off label" which I'll discuss below. What does "stage 2" mean? Type 1 diabetes moves through three stages. Stage 1 is when someone has two or more autoantibodies associated with T1D. Stage 2 is when they also have abnormally high blood glucose levels after meals. People in stage 2 don't have obvious symptoms, and so will not know their blood glucose levels are high unless they test. Stage 3 is when people start to have obvious symptoms of T1D. This is when T1D is usually diagnosed. TZIELD™/Teplizumab is not approved for people who have already been diagnosed with T1D or are in their honeymoon phase.
The treatment is a 14 day series of infusions. One infusion per day, and each one takes 30-60 minutes at a clinic. This is not an under the skin injection, nor can it be given by a random person at home, like insulin. Infusions require a trained professional working in a clinic.
In terms of "what will it do?" The official answer is, it will delay the onset of T1D symptoms (and therefore the need to inject insulin) for two years on average. However, the average delay may be longer than that. If you follow patients for 3 years, the longest delay you can measure is three years, but as you follow them longer, you might see a longer delay. The 2 year figure came from the original study (which followed people between 2 and 7 years (average of 4 1/4). Follow on data suggested that the the delay might be 3 years, based on following people longer. Therefore, as time moves forward and we continue to follow treated people, we might find that the average delay is even longer than the 3 years from the current data.
Teplizumab does have side effects. To quote the FDA: "The most common side effects of TZIELD include decreased levels of certain white blood cells, rash and headache." All of these resolved themselves soon after the infusions. Temporary Epstein–Barr virus reactivation was seen in one patient and has been seen occasionally in people who got other anti-CD3s treatments as part of organ transplantation. Obviously, anything that changes the immune system could have a long term side effect, but these have not been seen in people treated with Teplizumab.
Provention Bio (the makers of Teplizumab) has priced TZIELD at $193,000 (wholesale) to treat one person. That is not a typo. One hundred, ninety three thousand dollars US. Of course, that is the public price and it is not clear what insurance companies will really pay, or what the out of pocket will be for individuals.
This is from the company's press release:
Provention Bio has launched COMPASS, a patient support program with a
staff of dedicated personnel available to answer questions and help
navigate coverage, reimbursement and access for patients that are
prescribed TZIELD. Provention Bio offers financial assistance options
(e.g. copay assistance) to eligible patients for out-of-pocket costs.
Patients are encouraged to speak to their healthcare providers to find
out whether TZIELD is appropriate for them, and patients who have been
prescribed TZIELD and their healthcare providers can call
1-844-778-2246, Monday through Friday from 8AM-8PM EST or email a COMPASS Navigator at COMPASS@proventionbio.com.
Note: although this text makes it sound like only prescribed patients can use this facility, on their conference call, it was very clear that this phone number and email can be used by anyone who are interested in the treatment, even if they are not yet prescribed users. You can listen to Provention Bio's conference call here: https://app.webinar.net/nDvPqlJY9o8/on-demand
Should I Use It? Should My Child Use It?
You need to work with your medical team to answer this question.
This treatment is not targeted at people who have already been diagnosed with diabetes. Rather, it is focused on their siblings (and other first degree relatives). TZIELD is approved for people who test positive for two or more autoantibodies, and who have abnormal blood glucose levels after a meal, but are not yet seeing the symptoms of T1D (like thirst, excessive urination, weight loss, flu like symptoms, etc.) From a practical point of view, this makes siblings (and other first degree relatives) the first target of this treatment.
So the first thing to do, if you have not already done it, is to test siblings and other first degree relatives for autoantibodies, and then for dysglycemia.
JDRF's T1Detect program lists several ways to get tested for autoantibodies:
https://www.jdrf.org/t1d-resources/t1detect/
If a relative has T1D, then TrialNet will usually do it for free. Anyone can also order tests through their doctor, and there are free programs for some people in Colorado, South Dakota, and Washington state. In the past, I know a lot of people did not want to join TrialNet, because they thought "even if my child tests positive for autoantibodies, there is nothing I can do, so I would rather not know". Well, now there is something you can do. You can treat with TZIELD and delay the onset.
The next thing is to talk to your medical team about testing for dysglycemia, which is abnormally high blood glucose levels after a meal.
If you have two or more autoantibodies and dysglycemia, then you can talk to your medical team. This is a prescription drug, and only someone on the team can prescribe it, plus you need a clinic to actually administer it.
What Happens Next?
Provention believes that they will have supply to distribute by "the end of the year", which I interpret to be the end of 2022. They further believe that insurance will approve coverage for this drug in the next 6 or 9 months.
I have three thoughts on what is going to happen:
First, people are slowly going to start using TZIELD, which means that we are going to start learning what happens in actual use. There are two things to remember. The first is that drug approval is not like opening a faucet. It is not that thousands of people are going to suddenly start using the drug the day after it is approved (or even the first day it is available on the market). There is a long, slow time where patients and doctors become comfortable using the drug and recommending its use. The first question, which will take months or even years to answer is: will doctors recommend this and will people use it? Insurance coverage and real cost to people will matter as well. Once that is answered, we will move on to the second question, which is: are the benefits and risks the same as what was seen in the clinical testing? Just because a drug delayed the onset of T1D symptoms for 3 years in testing, does not mean it will do the same in real usage. Some drugs do better, some worse, and some the same. The same is true with side effects. They can be better or worse or the same as in testing.
Second, Provention Bio (the makers of Teplizumab) are going to try to get it approved for people in the honeymoon phase of T1D. That is, people in stage 3 of type 1 diabetes. Their PROTECT study is scheduled to finish in May 2023. If it does, and if it is successful, then use of Teplizumab use could expand after that. The PROTECT study is enrolling people in their honeymoon phase (within 6 weeks of diagnosis), who get two courses of therapy (so two different 14 infusion series, six months apart).
Provention Bio is also interested in getting TZIELD approved for people under 8 years old, and then later combining TZIELD with other treatments for a stronger response.
Third is off label use. In America, when a drug is approved, it is approved for a specific indication. An indication is a collection of symptoms that a patient has; both the patient and the symptoms are part of an indication. For example, TZIELD has been approved for people in stage 2 of type 1 diabetes (symptoms) who are 8 years old or older (patient). However, a doctor can prescribe any approved drug for any indication, not just what it was approved for. If they prescribe it for any other situation, that is called "off label use". This is not a legal grey area (although it sounds like one). It is perfectly legal.
There are some practical limitations to off label use. Insurance often will not pay for it, doctors are often reluctant to prescribe it, and the company can not talk about it at all. But the bottom line is that once TZIELD is available, a doctor could choose to prescribe it to someone who was in their honeymoon, had established T1D, or was younger than 8 years old. If this does happen, however, we might not hear about it. In general, the FDA forbids companies to mention any off label uses of their drugs. So even if one doctor notices that a particular off label use is successful, it is traditionally hard to communicate that to others. Although I suspect with modern social media, it is a lot easier than it used to be.
How Long Did It Take?
One of the key questions people ask me is, how long does it take the FDA to approve a cure for T1D? Up until now, the true answer to that question has been, since it has never happened, we don't know. In theory, it should take a drug about 15 years to work though the process of three phases of clinical trials and marketing approval as required by the FDA. But since it has never happened, no one knows for sure. That is all still true, of course, because Teplizumab is not a cure. But it is the closest we've gotten, so asking how long did it take is a reasonable question.
Teplizumab took a "wrong turn" on the way to approval, going through these four development phases:
- It took about 10 years to move into clinical trials. From the late 1980s to the late 1990s.
- It took at least 7 years to move from NOD mouse success to human trials (1992 to 1999)
- It then took about 11 years, from 1999 to 2010, to get from start of phase-I trials to the end of phase-III clinical trials. However, these trials were ultimately unsuccessful, and the company doing development dropped it.
- Clinical trials by TrialNet continued and the drug was picked up by another company. This second round of development took about 12 years from the previous failures though additional clinical trials and the marketing approval process, from 2010 to 2022.
In a broad sense, this supports my previous estimate that a drug can go from start of phase-I trials to market approval in about 15 years, if it is successful the first time. But the total research time for this treatment, from start of research (about 1986) to FDA approval (2022) was 36 years.
On a personal note, they started researching this about 15 years before my daughter was born and yet it is ready too late to help her, since she is now over 18 years into her established T1D.
Note: a longer, more detailed timeline is here:
https://cureresearch4type1diabetes.blogspot.com/2019/09/teplizumab-phase-ii-results-for.htmland there are more timeline dates in this article:
https://www.jdrf.org/blog/2022/11/17/fda-approves-tzield-teplizumab-watershed-moment-t1d-community/Why Did The First Round of Phase-III Clinical Trials Fail?
This is an important question. If the first round of clinical trials had been successful, then it is very reasonable to assume Teplizumab would have been approved 8 or even 10 years ago. A lot of people could have used it in that time period, so the question has real impact.
There are (at least) two answers to this question.
The first batch of research, which occurred between about 1999 and 2012 was trying to cure people who were in their honeymoon (stage 3) phase of T1D. It was the phase-III trial from this line of research that was unsuccessful.
However, there was a second batch of research, which was really just one clinical trial from 2010 to 2018, which was focused on delaying the onset of T1D during the "at risk" (stage 2) phase, and that was the research which ended up getting approved.
So two answers to the "why failed" question, are that the research that was unsuccessful was aiming "too high" at curing T1D, and the successful research was aimed lower, at delaying the onset of T1D. Also that the unsuccessful research was during the later stage 3 phase of T1D, but the successful research was during the earlier stage 2. But taken together, those two issues are not the whole story, because the phase-III study which was unsuccessful also used an unfortunate primary end point. This choice of end point might have led to the clinical trial being unsuccessful, even if the treatment later turns out to be successful. That brings up a discussion of end points and why they matter.
An end point is an official measure of success in a clinical trial. Clinical trials usually have one primary end point, a few secondary end points, and sometimes some tertiary end points. The primary end point determines success. It is what everyone looks at. For a phase-III clinical trial, it needs to measure the overall success of the treatment. It also has to be practical to measure, and meaningful to patients and doctors alike.
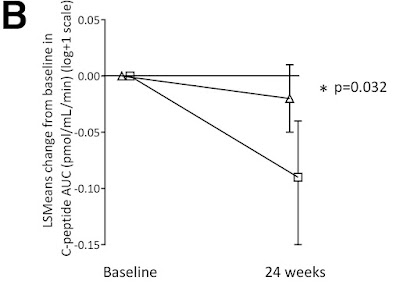
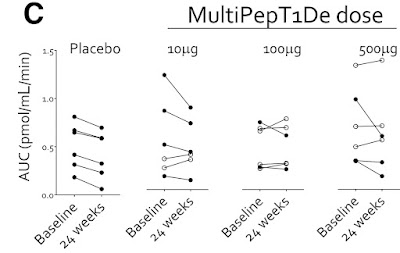
For the last 20 years, the primary end point for clinical trials aimed at curing type-1 diabetes has been C-peptide levels. C-peptide has many advantages as an end point: it is easy and reliable to measure, and it accurately reflects how much insulin a person's body is generating, separate from how much they are injecting. Even if the person is not completely cured, C-peptides measure progress towards the cure. It is a good primary outcome. But this was not always known. Consensus that measuring C-peptides was the best primary outcome occurred in the 2001-2004 time frame.
The big phase-III trial (which ended up failing) used a combination of insulin use and A1c (not C-peptides) as a primary end point. In retrospect, that might have been a mistake. Provention Bio is currently running a phase-III trial testing Teplizumab's ability to help people in the honeymoon phase, and they are using C-peptide as the primary end point. If this newer trial is successful, it will be clear that choice of end points had a big impact.
A related question is why didn't the earlier researchers focus more on delaying T1D and less on curing it, so they could have been successful earlier? That is easy to answer: it was not possible to research treatments which might delay the onset of T1D, until TrialNet's landmark Pathway to Prevention research. That started in 2004 and took several years to gather enough data so that it was understood what normal progression of T1D was, or how to measure it. Prior to that, there was simply no way to even evaluate a treatment which might delay onset.
Who Should We Thank?
Mythology requires heroes, so everyone wants to point at one person (or maybe two) and say they are the heroes. They did it. But real science rarely works that way. There are always a lot of different people bringing a lot of different skills to the party.
If you want to go the mythical hero route, then I suggest Jeffery A Bluestone and Kevan C Herold, as your heroes. They collaborated in the development of Teplizumab from the very earliest research to the most recent clinical trials and corporate sponsorship. Also, they pursued this research even in the face of setbacks, and ended up overcoming all the issues.
On the other hand, if you want a more realistic group, reflecting the large number of people required to develop an immunological treatment, the long history of this treatment, and the many factors that came together to lead to its success, then add:
- All the researchers who worked on anti-CD3 based treatments for T1D in both animals and people. Collectively, they authored dozens of papers which led to the treatment we have today. It would be an interesting project to get every name on every paper. My guess is that there would be over 100 researchers in the list, and that doesn't cover the people who contributed in ways that did not end up as paper authors. As part of this blog, I tried to find out who was the lead author of the first paper that showed an anti-CD3 worked against T1D in animals. My two leading contenders are Anthony R. Hayward and Lucienne Chatenoud, but there may be even earlier examples.
- All the patients who participated in the 14 clinical studies on Teplizumab, and all the researchers who ran them. FDA approval is based on data, and these studies created the data.
- The JDRF, for several contributions. First, is the money. They funded Teplizumab throughout its development (roughly 35 years), from the earliest pre-clinical research, through the clinical trials, and right up to providing venture capital style funding to Provention Bio through their T1D Fund. Money matters. Second, JDRF was part of the T1D community which pushed the use of C-peptides as the best primary end point, and also the "stages" view of the natural history of T1D. Both of these scientific policies were critical to the eventual success of Teplizumab.
- TrialNet for two different reasons. First, their "Pathway to Prevention" trial completely changed how we thought about the onset of T1D, leading to the "stages" model. TrialNet gave us the data foundational to the very idea of delaying the onset. Second, they sponsored the one clinical trial (called TN-10) which succeeded, and they did it after commercially sponsored trials ended unsuccessfully. This shows some "guts".
- Provantage Bio, which invested money in a drug that had already failed
previous phase-III clinical trials. They had the foresight to try
again, and were successful. This is something that rarely happens in
the world of pharmaceuticals.
More About Teplizumab / TZIELD
Teplizumab is a humanized monoclonal antibody which targets CD3 cells in the immune system in order to lower (or stop) the body's autoimmune response. This treatment tries to prevent type-1, or lessen it's severity, by "turning down" the immune system's attack on the body's own pancreas cells. This basic approach has resulted in treatments (but not cures) for other autoimmune diseases. This is a highly targeted approach; it is not lowering the whole immune system, but rather a specific part involved in T1D.
Monoclonal antibodies are therapies created by cloning a single cell that attacks the kind of cell you don't want. You end up with a vast number of identical cells, all of which attack the kind of cell you don't want. By carefully choosing the starting cell, you can "target" the monoclonal antibody to attack a very specific type of immune cell. Because of how they are produced, early monoclonal antibodies were targeted at mouse cells, rather than human cells, and this sometimes caused problems. Later techniques were developed to make the cells more human like, but still not completely human. These were called "humanized" or "chimeric" monoclonal antibodies. Finally, techniques were developed to create fully human monoclonal antibodies, and these are called "human" or "fully humanized". Teplizumab is a fully humanized monoclonal antibody.
There are scores of monoclonal antibodies (of all three types) approved for use in the United States, for a wide variety of illnesses. Different monoclonal antibodies have different safety profiles. Most have names ending in "mab".
More Reading
Pricing information comes from these articles:
https://endpts.com/provention-gets-fda-nod-on-drug-to-delay-type-i-diabetes-kicking-off-us-launch-with-sanofi/
C-peptide as the best primary end point:
https://pubmed.ncbi.nlm.nih.gov/14693724/
Presentation on why Teplizumab works:
https://www.easd.org/media-centre/home.html#!resources/studies-of-the-mechanism-of-teplizumab
Official press release:
https://investors.proventionbio.com/2022-11-17-TZIELD-TM-teplizumab-mzwv-approved-by-FDA-as-the-first-and-only-treatment-indicated-to-delay-the-onset-of-Stage-3-type-1-diabetes-T1D-in-adult-and-pediatric-patients-aged-8-years-and-older-with-Stage-2-T1D
https://medicine.yale.edu/news-article/fda-approves-first-drug-that-delays-onset-of-type-1-diabetes/
These are the earliest successful anti-CD3 in NOD mouse studies I could find:
https://pubmed.ncbi.nlm.nih.gov/1532716/
https://pubmed.ncbi.nlm.nih.gov/8278351/
Historical read:
https://endpts.com/we-kept-at-it-jeffrey-bluestone-plots-late-stage-comeback-after-teplizumab-shown-to-delay-type-1-diabetes/
End Notes
I refer to Teplizumab as a drug throughout this posting. Teplizumab is actually a biologic. Drugs are chemicals, while biologics are biological material. Teplizumab is a very specific type of cell. The FDA has separate sets of regulations for drugs vs. biologics. However, I feel using the word "drug" makes the blog more understandable to more people.
The product's name is TZIELD™ in Provention Bio's marketing literature. At least what I saw. However, it is Tzield™ on the bottle. I don't know which is correct, so I'm using all caps.
TZIELD is pronouced "TEE-zield" the second part is like "shield" but with a z sound. The International Phonetic Alphabet form is /tizil̩d/ or /tiziəld/.
Provention Bio is a publicly traded company, meaning you can buy and sell stock right now. Therefore, there are a bunch of financial websites and stock news service articles on the company as an investment opportunity. (I don't have an opinion on that.) However, reading these articles does give a completely different view of the company and product. We care about: does it work? They care about: how profitable will it be?
Joshua Levy
http://cureresearch4type1diabetes.blogspot.com
publicjoshualevy at gmail dot com
All
the views expressed here are those of Joshua Levy, and nothing here is
official JDRF or JDCA news, views, policies or opinions. My daughter has
type-1 diabetes and participates in clinical trials, which might be
discussed here. My blog contains a more complete non-conflict of
interest statement. Thanks to everyone who helps with the blog.